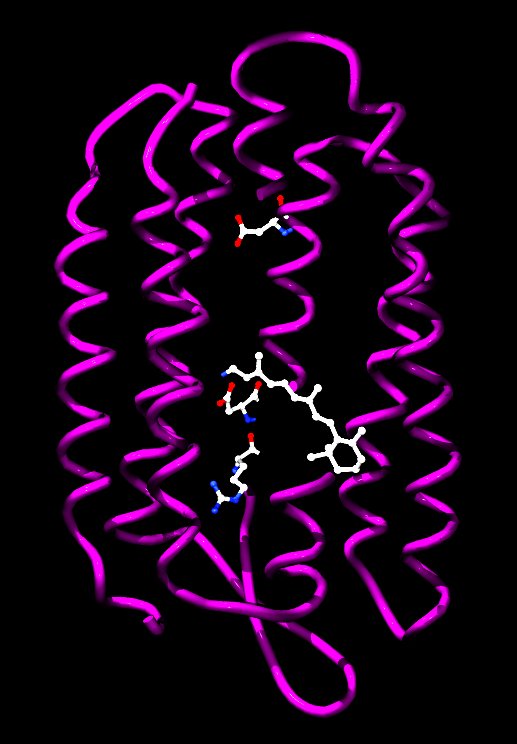
Bacteriorhodopsin is a protein used by Archaea, most notably Halobacteria (Domain Archaea, Kingdom Euryarchaeota, Phylum Euryarchaeota, Class Halobacteria). It acts as a proton pump, i.e. it captures light energy and uses it to move protons across the membrane out of the cell. The resulting proton gradient is subsequently converted into chemical energy. It is the retinal molecule that changes its conformation when absorbing a photon, resulting in a conformational change of the surrounding protein and the proton pumping action. Halobacteria (also Halomebacteria) are found in water saturated or nearly saturated with salt. They are also called halophiles, though this name is also used for other organisms that live in somewhat less concentrated salt water. They are common in most environments where large amounts of salt, moisture, and organic material are available. Halobacteria can grow aerobically or anaerobically. Parts of the membranes of halobacteria are purplish in color, and large blooms of Halobacteria appear reddish, from the pigment bacteriorhodopsin, related to the retinal pigment rhodopsin. This pigment is used to absorb light, which provides energy to create Adenosine triphosphate (ATP). Halobacteria also possess a second pigment, halorhodopsin, which pumps chloride ions in the cell in response to photons, creating a voltage gradient and assisting in the production of energy from light. The process is unrelated to other forms of photosynthesis involving electron transport however, and halobacteria are incapable of fixing carbon from carbon dioxide. Archaea evolve extremely slowly, and thus this modern bacteriorhodopsin is likely little changed from deep time on earth. |